Decision tree

(Later: add curved-arrow mechanism diagrams.)
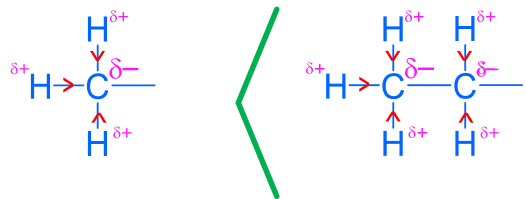
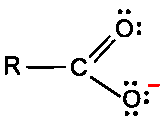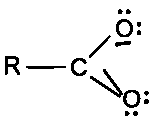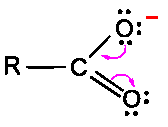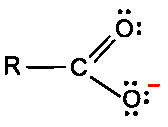
Carbonyl C=O is polarized: C is electrophilic, O is nucleophilic.
- Nucleophilic addition to aldehydes/ketones
- Acyl substitution for carboxylic derivatives
Carbonyl carbon is electrophilic due to C=O polarization; nucleophiles attack the carbonyl carbon.
| Reaction | Reagent | Product / Key feature |
|---|---|---|
| Hydride reduction (mild) | NaBH₄ | Aldehyde → 1° alcohol; Ketone → 2° alcohol |
| Hydride reduction (strong) | LiAlH₄ | Reduces aldehydes, ketones, esters, acids → alcohols |
| Cyanohydrin formation | HCN | H⁺ and C≡N⁻ added; forms cyanohydrin RCH(OH)CN |
| Acetal / ketal formation | ROH + acid | Protecting group; forms acetal / ketal (reversible) |
| Imine formation | RNH₂ + acid | C=N (imine); condensation reaction |
| Grignard addition | RMgBr (then H₃O⁺) | New C–C bond; alcohol formed after workup |
| Acyl substitution | Acid chloride / ester + Nu⁻ | Addition–elimination (nucleophilic acyl substitution) |
Predict carbonyl reaction
(Later: add curved-arrow stepper with tetrahedral intermediate.)
SR (radical substitution) proceeds via a radical chain mechanism: initiation → propagation → termination.
- Typical: Alkane halogenation (Cl₂ / Br₂, hν or Δ)
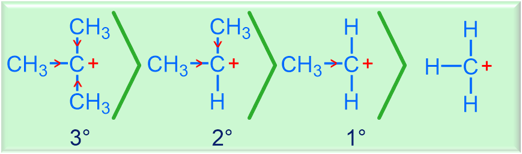
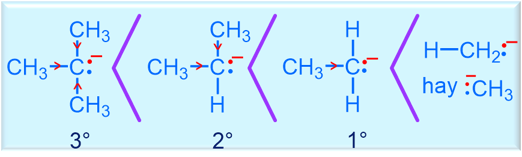
- Key factors: radical stability & selectivity (Br₂ more selective than Cl₂)
- Outcome: mixture of products; controlled by relative H-abstraction rates
| Feature | Description |
|---|---|
| Typical substrate | Alkanes (sp³ C–H bonds) |
| Reagents | Cl₂ or Br₂ + light (hν) or heat (Δ) |
| Mechanism type | Free radical chain reaction |
| Steps |
1) Initiation (radical formation) 2) Propagation (chain continues) 3) Termination (radicals combine) |
| Selectivity |
Br₂ is more selective than Cl₂ (3° > 2° > 1°) |
| Major product rule | Hydrogen is replaced at the position that forms the most stable radical |
| Initiation | Cl₂ → 2 Cl• (homolytic cleavage) |
| Propagation (step 1) |
Cl• + R–H → HCl + R• |
| Propagation (step 2) |
R• + Cl₂ → R–Cl + Cl• |
| Termination |
Cl• + Cl• → Cl₂ R• + Cl• → R–Cl R• + R• → R–R |
Radical halogenation is thermodynamically driven by bond strengths and kinetically influenced by radical stability.
Predict Radical Substitution Product Distribution
| Type | Count |
|---|---|
| 1° (primary) | |
| 2° (secondary) | |
| 3° (tertiary) |
(Later: add an interactive “which H is substituted?” selector + selectivity chart.)
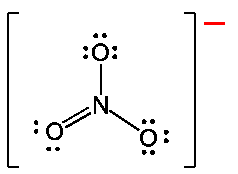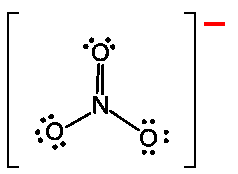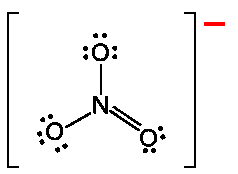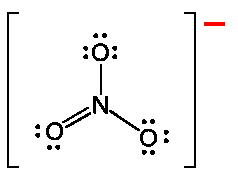
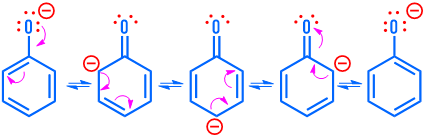
Electrophilic Aromatic Substitution (EAS) preserves aromaticity. An electrophile replaces a hydrogen on the benzene ring.
| Step 1 | Formation of strong electrophile (E⁺) |
| Step 2 | Aromatic ring attacks E⁺ → σ-complex (arenium ion) |
| Step 3 | Deprotonation restores aromaticity |
The σ-complex (arenium ion) is resonance-stabilized but temporarily loses aromaticity.
| Reaction | Reagents | Electrophile |
|---|---|---|
| Halogenation | Br₂ / FeBr₃ or Cl₂ / FeCl₃ | Br⁺ or Cl⁺ |
| Nitration | HNO₃ / H₂SO₄ | NO₂⁺ (nitronium) |
| Sulfonation | SO₃ / H₂SO₄ | SO₃ or HSO₃⁺ |
| Friedel–Crafts alkylation | R–Cl / AlCl₃ | R⁺ (carbocation) |
| Friedel–Crafts acylation | RCOCl / AlCl₃ | RCO⁺ (acylium) |
| Substituent Type | Examples | Direction | Activation |
|---|---|---|---|
| Electron-donating (EDG) | –OH, –OR, –NH₂, –R | Ortho / Para | Activating |
| Weakly deactivating halogens | –Cl, –Br | Ortho / Para | Deactivating |
| Electron-withdrawing (EWG) | –NO₂, –CF₃, –CN, –COOH | Meta | Deactivating |
Rule of thumb:
• EDG → stabilize σ-complex → ortho/para
• Strong EWG → destabilize ortho/para σ-complex → meta
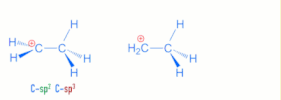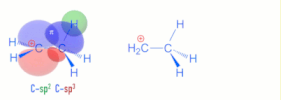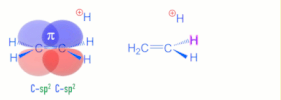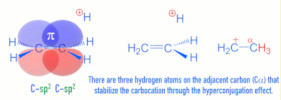




 >
>
